Optimizing DNA Extraction Methods from Leaf and Wood Tissues to Support Dipterocarp Conservation and Sustainable Forest Management
Abstract
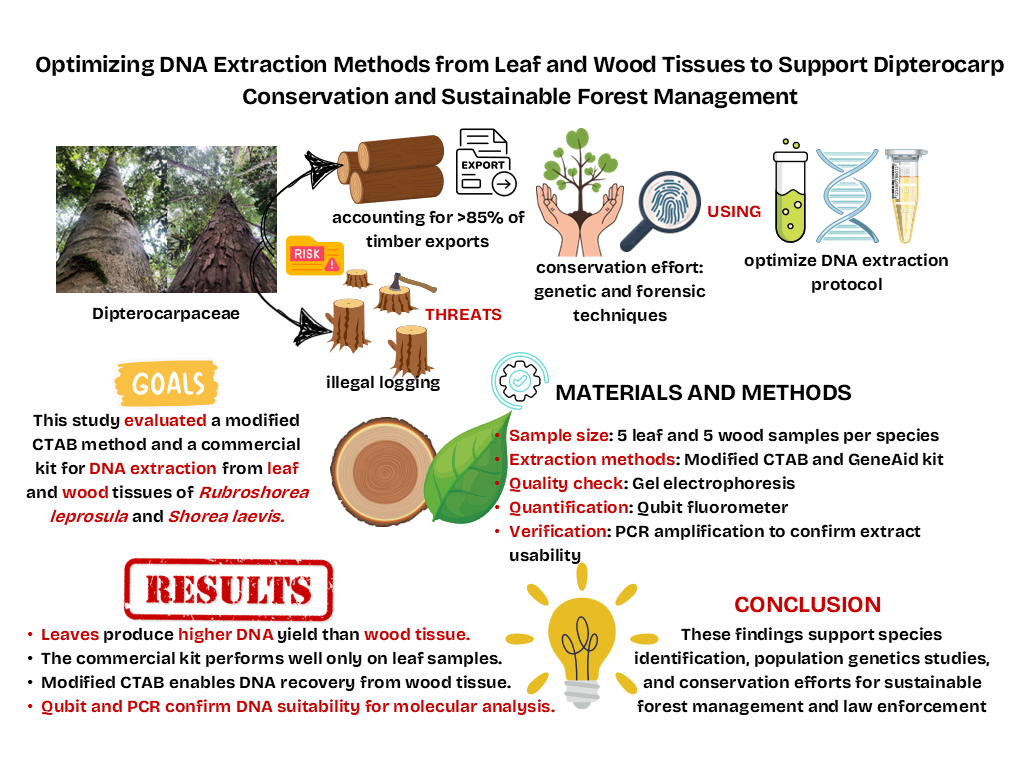
Dipterocarpaceae are economically important, contributing over 85% of Indonesia's timber exports. However, this crucial resource is increasingly threatened by illegal logging, habitat destruction, and the illegal timber trade, which jeopardize dipterocarp population. Furthermore, conservation efforts utilizing genetic and forensic techniques often encounter substantial challenges due to the complexities in DNA extraction protocol. To address this, the study aimed to enhance the efficiency of DNA extraction methodologies by comparing two methods: the modified cetyltrimethylammonium bromide (CTAB) and the Genomic DNA Mini Kit (Plant) from Geneaid Biotech Ltd. The research focused on leaf and wood samples from two species, spesifically Rubroshorea leprosula (Miq.) P.S.Ashton & J.Heck and Shorea laevis Ridl. For each of these species, five leaf and five wood samples were extracted using both methods. The quality of the DNA extraction was evaluated using electrophoresis and quantified with a Qubit fluorometer. Higher DNA concentrations were obtained with the modified CTAB method compared to the GeneAid kit for both R. leprosula and S. laevis, particularly in leaf tissue. The GeneAid kit consistently exhibited low DNA yield efficiency compared to the modified CTAB method for both species. Additionally, PCR amplification of both leaf and wood samples confirmed that the extracted DNA was suitable for
molecular analyses. These findings not only contributed to laboratory applications but also served as practical tools for species identification and genetic conservation for sustainable forest management and law enforcement.
Full text article
References
1. Symington, C.. Foresters’ Manual of Dipterocarps; University of Malaya Press: Malayan Forest Records, 1943;
2. Widiyatno; Indrioko, S.; Na’iem, M.; Uchiyama, K.; Numata, S.; Ohtani, M.; Matsumoto, A.; Tsumura, Y. Effects of Different Silvicultural Systems on the Genetic Diversity of Shorea parvifolia Populations in the Tropical Rainforest of Southeast Asia. Tree Genetics and Genomes 2016, 12, doi:10.1007/s11295-016-1030-y.
3. Ghazoul, J. Dipterocarp Biology, Ecology, and Conservation; Oxford University Press, 2016;
4. Ashton, P.S. Dipterocarpaceae. Tree Flora of Sabah and Sarawak 2004, 222, 63–337, doi:10.26525/tfss5002.
5. Maycock, C.R.; Kettle, C.J.; Khoo, E.; Pereira, J.T.; Sugau, J.B.; Nilus, R.; Ong, R.C.; Amaludin, N.A.; Newman, M.F.; Burslem, D.F.R.P. A Revised Conservation Assessment of Dipterocarps in Sabah. Biotropica 2012, 44, 649–657, doi:10.1111/j.1744-7429.2011.00852.x.
6. Luo, W.; Strijk, J.S.; Barstow, M.; Wee, A.K.S. The Role of Protected Areas in Tropical Tree Conservation Post-2020: A Case Study Using Threatened Dipterocarpaceae. Biological Conservation 2022, 272, 109634, doi:10.1016/j.biocon.2022.109634.
7. Ministry of Environment and Forestry (MOEF) State of Indonesia’s Forest 2018; Jakarta, 2018;
8. Statistics Indonesia (BPS) Statistical Yearbook of Indonesia 2021 Available online: https://www.bps.go.id.
9. Bartholomew, D.; Barstow, M.; Randi, A.; Bodos, V.; Cicuzza, D.; Kiat Hoo, P.; Juiling, S.; Khoo, E.; Kusumadewi, Y.; Majapun, R.; et al. The Red List of Bornean Endemic Dipterocarps GTA Global Tree Assessment; Botanic Gardens Conservation Intern: Richmond, UK, 2021; ISBN 1905164793.
10. Khoo, E.; Barstow, M.; Maycock, C.; Bodos, V.; Chong, K.Y.; Chua, L.S.L.; Cicuzza, D.; Deepu, S.; Dhyani, A.; Divina, A.N.; et al. The Red List of Dipterocarpaceae; Botanic Gardens Conservation International: Richmond, UK, 2022; ISBN 9781905164820.
11. Ashton, P.S. Dipterocarpaceae. In Flora Malesiana, I, Spermatophyta; Rijksherbarium: Leiden, 1982; pp. 237–552 ISBN 9789024726967.
12. Dwiyanti, F.G.; Harada, K.; Siregar, I.Z.; Kamiya, K. Population Genetics of the Critically Endangered Species Dipterocarpus littoralis Blume (Dipterocarpaceae) Endemic in Nusakambangan Island, Indonesia. Biotropia 2014, 21, 1–12, doi:10.11598/btb.2014.21.1.1.
13. Kusuma, Y.W.C.; Surya, M.I.; Kurniawati, S.; Yulita, K.D.S.; Destri; Risna, R.A.; Sudarmonowati, E.; Matsuo, A.; Kurita, K.; Suyama, Y.; et al. Genetic Diversity and Structure of Hopea bilitonensis, an Endemic Dipterocarp from Belitung Island, Indonesia. Journal of Asia-Pacific Biodiversity 2024, 17, 400–405, doi:10.1016/j.japb.2024.01.008.
14. Rachmat, H.H.; Hidayat, A.; Kamiya, K.; Harada, K. Revealing the Genetic Structure of Indonesia Moluccan Dipterocarps Species, Rubroshorea selanica. International Journal of Agriculture and Biology 2024, 31, 183–191, doi:10.17957/IJAB/15.2130.
15. Widiyono, W. Biological and Economic Value of Dipterocarpaceae, the Main Timber Forest Product of Indonesia. Indonesian Journal of Applied Environmental Studies 2021, 1, 104–112.
16. Zulfahmi; Mahfira, U.O.; Siregar, U.J.; Siregar, I.Z.; Yunanto, T. Comparison of Levels of Chloroplast DNA Diversity of Two Shorea Species with Contrasting Geographical Distribution. Asia-Pacific Journal of Molecular Biology and Biotechnology 2015, 23, 291–302, doi:10.21307/APJMBB-2015-009.
17. Susilowati, A.; Wijaya, K.; Mawazin; Rachmat, H.H.; Lismayati, Y.; Kurniawan, H.; Ginting, I.M. Conserving Dipterocarps Biodiversity in Remnant Forests of Small Islands in Batam Island, Indonesia. Biodiversitas 2024, 25, 726–733, doi:10.13057/biodiv/d250232.
18. Tsumura, Y.; Kado, T.; Yoshida, K.; Tani, N. Molecular Database for Classifying Shorea Species (Dipterocarpaceae) and Techniques for Checking the Legitimacy of Timber and Wood Products. Journal of Plant Research 2011, 124, 35–48, doi:10.1007/s10265-010-0348-z.
19. Hiroshi, A.; Nakai, Y.; Hasegawa, G.P. Economic Importance of the Endemic Sumatran Lowland Dipterocarp Tree Species (Shorea javanica). Afr J Trop Agric 2015, 3, 163–172.
20. Gasson, P.E.; Lancaster, C.A.; Young, R.; Redstone, S.; Miles-Bunch, I.A.; Rees, G.; Guillery, R.P.; Parker-Forney, M.; Lebow, E.T. WorldForestID: Addressing the Need for Standardized Wood Reference Collections to Support Authentication Analysis Technologies; a Way Forward for Checking the Origin and Identity of Traded Timber. Plants People Planet 2021, 3, 130–141, doi:10.1002/ppp3.10164.
21. Dormontt, E.E.; Boner, M.; Braun, B.; Breulmann, G.; Degen, B.; Espinoza, E.; Gardner, S.; Guillery, P.; Hermanson, J.C.; Koch, G.; et al. Forensic Timber Identification: It’s Time to Integrate Disciplines to Combat Illegal Logging. Biological Conservation 2015, 191, 790–798, doi:10.1016/j.biocon.2015.06.038.
22. Luo, L.; Ye, P.; Lin, Q.; Liu, M.; Hao, G.; Wei, T.; Sahu, S.K. From Sequences to Sustainability: Exploring Dipterocarp Genomes for Oleoresin Production, Timber Quality, and Conservation. Plant Science 2024, 346, 112139, doi:10.1016/j.plantsci.2024.112139.
23. Fatima, T.; Srivastava, A.; Hanur, V.S.; Rao, M.S. An Effective Wood DNA Extraction Protocol for Three Economic Important Timber Species of India. American Journal of Plant Sciences 2018, 09, 139–149, doi:10.4236/ajps.2018.92012.
24. Tanis, M.H.; Wallberg, O.; Galbe, M.; Al-Rudainy, B. Lignin Extraction by Using Two-Step Fractionation: A Review. Molecules 2024, 29, doi:10.3390/molecules29010098.
25. Liepelt, S.; Sperisen, C.; Deguilloux, M.F.; Petit, R.J.; Kissling, R.; Spencer, M.; De Beaulieu, J.L.; Taberlet, P.; Gielly, L.; Ziegenhagen, B. Authenticated DNA from Ancient Wood Remains. Annals of Botany 2006, 98, 1107–1111, doi:10.1093/aob/mcl188.
26. Swetha, V.P.; Parvathy, V.A.; Sheeja, T.E.; Sasikumar, B. Isolation and Amplification of Genomic DNA from Barks of Cinnamomum Spp. Turkish Journal of Biology 2014, 38, 151–155, doi:10.3906/biy-1308-5.
27. Abe, H.; Watanabe, U.; Yoshida, K.; Kuroda, K.; Zhang, C. Changes in Organelle and DNA Quality, Quantity, and Distribution in the Wood of Cryptomeria japonica over Long-Term Storage. IAWA Journal 2011, 32, 263–272.
28. Deguilloux, M.F.; Pemonge, M.H.; Petit, R.J. Novel Perspectives in Wood Certification and Forensics: Dry Wood as a Source of DNA. Proceedings of the Royal Society B: Biological Sciences 2002, 269, 1039–1046, doi:10.1098/rspb.2002.1982.
29. Marsal, G.; Boronat, N.; Canals, J.M.; Zamora, F.; Fort, F. Comparison of the Efficiency of Some of the Most Usual DNA Extraction Methods for Woody Plants in Different Tissues of Vitis vinifera L. Journal International des Sciences de la Vigne et du Vin 2013, 47, 227–237, doi:10.20870/oeno-one.2013.47.4.1559.
30. Jiao, L.; Lu, Y.; He, T.; Guo, J.; Yin, Y. DNA Barcoding for Wood Identification: Global Review of the Last Decade and Future Perspective. IAWA Journal 2020, 41, 620–643, doi:10.1163/22941932-bja10041.
31. Ng, C.H.; Ng, K.K.S.; Lee, S.L.; Zakaria, N.F.; Lee, C.T.; Tnah, L.H. DNA Databases of an Important Tropical Timber Tree Species Shorea leprosula (Dipterocarpaceae) for Forensic Timber Identification. Scientific Reports 2022, 12, 1–11, doi:10.1038/s41598-022-13697-x.
32. Gan, K.S.; Lim, S.C. Common Commercial Timbers of Peninsular Malaysia. Research Pamphlet - Forest Research Institute Malaysia 2004, xviii + 130 pp.
33. Tnah, L.H.; Lee, S.L.; Ng, K.K.S.; Bhassu, S.; Othman, R.Y. DNA Extraction from Dry Wood of Neobalanocarpus heimii (Dipterocarpaceae) for Forensic DNA Profiling and Timber Tracking. Wood Science and Technology 2012, 46, 813–825, doi:10.1007/s00226-011-0447-6.
34. Doyle, J.J.; Doyle, J.L. A Rapid DNA Isolation Procedure for Small Quantities of Fresh Leaf Tissue. Phytochemical Bulletin 1987, 19, 11–15.
35. Yulita, K.S.; Bayer, R.J.; West, J.G. Molecular Phylogenetic Study of Hopea and Shorea (Dipterocarpaceae): Evidence from the trnL-trnF and Internal Transcribed Spacer Regions. Plant species biology 2005, 20, 167—182, doi:10.1111/j.1442-1984.2005.00136.x.
36. Harnelly, E.; Thomy, Z.; Fathiya, N. Phylogenetic Analysis of Dipterocarpaceae in Ketambe Research Station, Gunung Leuser National Park (Sumatra, Indonesia) Based on rbcL and matK Genes. Biodiversitas 2018, 19, 1074–1080, doi:10.13057/biodiv/d190340.
37. Indrioko, S.; Gailing, O.; Finkeldey, R. Molecular Phylogeny of Dipterocarpaceae in Indonesia Based on Chloroplast DNA. Plant Systematics and Evolution 2006, 261, 99–115, doi:10.1007/s00606-006-0435-8.
38. Rachmat, H.H.; Yulita, K.S.; Dwiyanti, F.G.; Susilowati, A.; Arrofaha, N.; Susila, S.; Kamal, I.; Siregar, I.Z. Optimizing DNA Extraction and Selecting Suitable Regions for Biodiversity Assessment: A Study on Shorea leprosula. Jurnal Manajemen Hutan Tropika 2024, 30, 144–154, doi:10.7226/jtfm.30.1.144.
39. Moura, C.C.D.M.; Brambach, F.; Jair, K.; Bado, H.; Krutovsky, K. V; Kreft, H.; Tjitrosoedirdjo, S.S.; Siregar, I.Z.; Gailing, O. Integrating DNA Barcoding and Traditional Taxonomy for the Identification of Dipterocarps in Remnant Lowland Forests of Sumatra. Plants 2019, 8, 1–14.
40. Taberlet, P.; Gielly, L.; Pautou, G.; Bouvet, J. Universal Primers for Amplification of Three Non-Coding Regions of Chloroplast DNA. Plant Molecular Biology 1991, 17, 1105–1109, doi:10.1007/BF00037152.
41. Shioya, N.; Ogiso-tanaka, E.; Watanabe, M.; Anai, T.; Hoshino, T. Development of a High-Quality / Yield Long-Read. Plants 2023, 12, 1–14.
42. Moreira, P.A.; Oliveira, D.A. Leaf Age Affects the Quality of DNA Extracted from Dimorphandra mollis (Fabaceae), a Tropical Tree Species from the Cerrado Region of Brazil. Genetics and molecular research : GMR 2011, 10, 353–358, doi:10.4238/vol10-1gmr1030.
43. Barta, C.E.; Bolander, B.; Bilby, S.R.; Brown, J.H.; Brown, R.N.; Duryee, A.M.; Edelman, D.R.; Gray, C.E.; Gossett, C.; Haddock, A.G.; et al. In Situ Dark Adaptation Enhances the Efficiency of DNA Extraction from Mature Pin Oak (Quercus palustris) Leaves, Facilitating the Identification of Partial Sequences of the 18S RRNA and Isoprene Synthase (IspS) Genes. Plants 2017, 6, doi:10.3390/plants6040052.
44. Stettinius, A.; Holmes, H.; Zhang, Q.; Mehochko, I.; Winters, M.; Hutchison, R.; Maxwell, A.; Holliday, J.; Vlaisavljevich, E. DNA Release from Plant Tissue Using Focused Ultrasound Extraction (FUSE). Applications in Plant Sciences 2023, 11, 1–13, doi:10.1002/aps3.11510.
45. Gao, C.; Cui, X.; Matsumura, J. Multidimensional Exploration of Wood Extractives: A Review of Compositional Analysis, Decay Resistance, Light Stability, and Staining Applications; 2024; Vol. 15; ISBN 8615900302035.
46. Nawrot-Chorabik, K.; Sułkowska, M.; Gumulak, N. Secondary Metabolites Produced by Trees and Fungi: Achievements So Far and Challenges Remaining. Forests 2022, 13, 1–31, doi:10.3390/f13081338.
47. Aboul-Maaty, N.A.-F.; Oraby, H.A.-S. Extraction of High-Quality Genomic DNA from Different Plant Orders Applying a Modified CTAB-Based Method. Bulletin of the National Research Centre 2019, 43, 1–10, doi:10.1186/s42269-019-0066-1.
48. Sahu, S.K.; Thangaraj, M.; Kathiresan, K. DNA Extraction Protocol for Plants with High Levels of Secondary Metabolites and Polysaccharides without Using Liquid Nitrogen and Phenol. ISRN Molecular Biology 2012, 2012, 1–6, doi:10.5402/2012/205049.
49. Khanuja, S.P.S.; Shasany, A.K.; Darokar, M.P.; Kumar, S. Rapid Isolation of DNA from Dry and Fresh Samples of Plants Producing Large Amounts of Secondary Metabolites and Essential Oils. Plant Molecular Biology Reporter 1999, 17, 74, doi:10.1023/A:1007528101452.
50. Nguyen, D.M.; Nguyen, H.L.P.; Nguyen, T.M. Genetic Structure of the Endemic Dipterocarpus condorensis Revealed by Microsatellite Markers. AoB PLANTS 2022, 14, 1–9, doi:10.1093/aobpla/plac007.
51. Walters, S.J.; Robinson, T.P.; Byrne, M.; Nevill, P. Seed Sourcing in the Genomics Era: Multispecies Provenance Delineation for Current and Future Climates. Restoration Ecology 2022, 30, 1–14, doi:10.1111/rec.13718.
52. Yu, M.; Jiao, L.; Guo, J.; Wiedenhoeft, A.C.; He, T.; Jiang, X.; Yin, Y. DNA Barcoding of Vouchered Xylarium Wood Specimens of Nine Endangered Dalbergia Species. Planta 2017, 246, 1165–1176, doi:10.1007/s00425-017-2758-9.
53. Lowe, A.J.; Wong, K.N.; Tiong, Y.S.; Iyerh, S.; Chew, F.T. A DNA Method to Verify the Integrity of Timber Supply Chains; Confirming the Legal Sourcing of Merbau Timber from Logging Concession to Sawmill. Silvae Genetica 2010, 59, 263–268, doi:10.1515/sg-2010-0037.
54. Malek, E.J.; Abdul Rahim, A.R. A Thematic Review of Forest Certification Publications from 2017 to 2021: Analysis of Pattern and Trends for Future Studies. Trees, Forests and People 2022, 10, 100331, doi:10.1016/j.tfp.2022.100331.
55. Susilawati, D.; Kanowski, P.J. Sustainability Certification and Legality Verification in Indonesian Natural Forest-Based Wood Products Value Chains. International Forestry Review 2021, 23, 365–391, doi:10.1505/146554821833992839.
56. Ng, C.H.; Ng, K.K.S.; Lee, S.L.; Tnah, L.H.; Lee, C.T.; Zakaria, N.F. A Geographical Traceability System for Merbau (Intsia palembanica Miq.), an Important Timber Species from Peninsular Malaysia. Forensic Science International: Genetics 2020, 44, 102188, doi:10.1016/j.fsigen.2019.102188.
57. Tang, X.; Zhao, G.; Ping, L. Wood Identification with PCR Targeting Noncoding Chloroplast DNA. Plant Molecular Biology 2011, 77, 609–617, doi:10.1007/s11103-011-9837-2.
58. Finkeldey, R.; Leinemann, L.; Gailing, O. Molecular Genetic Tools to Infer the Origin of Forest Plants and Wood. Applied Microbiology and Biotechnology 2010, 85, 1251–1258, doi:10.1007/s00253-009-2328-6.
59. Zeng, Y.; Wu, Z.; Zhang, C.; Meng, Z.; Jiang, Z.; Zhang, J. DNA Barcoding of Mobulid Ray Gill Rakers for Implementing CITES on Elasmobranch in China. Scientific Reports 2016, 6, 1–9, doi:10.1038/srep37567.
60. Akhmetzyanov, L.; Copini, P.; Sass-Klaassen, U.; Schroeder, H.; de Groot, G.A.; Laros, I.; Daly, A. DNA of Centuries-Old Timber Can Reveal Its Origin. Scientific Reports 2020, 10, 1–11, doi:10.1038/s41598-020-77387-2.
61. Tnah, L.H.; Lee, S.L.; Ng, K.K.S.; Faridah, Q.Z.; Faridah-Hanum, I. Forensic DNA Profiling of Tropical Timber Species in Peninsular Malaysia. Forest Ecology and Management 2010, 259, 1436–1446, doi:10.1016/j.foreco.2010.01.017.
62. Cetera, K.; Said, Z.; Dellarose Boer, F.; Nurul Qomariah, I.; Suprapto, E.; Triyanto, S. How Wood Identification Technologies Help Ensure Timber Legality in Indonesia. World Resources Institute 2021, 1–32, doi:10.46830/wriwp.18.00084.
63. Lowe, A.J.; Cross, H.B. The Application of DNA Methods to Timber Tracking and Origin Verification. IAWA Journal 2011, 32, 251–262, doi:10.1163/22941932-90000055.
64. Obayashi, K.; Tsumura, Y.; Ihara-Ujino, T.; Niiyama, K.; Tanouchi, H.; Suyama, Y.; Washitani, I.; Lee, C.T.; Lee, S.L.; Muhammad, N. Genetic Diversity and Outcrossing Rate between Undisturbed and Selectively Logged Forests of Shorea curtisii (Dipterocarpaceae) Using Microsatellite DNA Analysis. International Journal of Plant Sciences 2002, 163, 151–158, doi:10.1086/324549.
65. Tam, N.M.; Duy, V.D.; Duc, N.M.; Giap, V.D.; Xuan, B.T.T. Genetic Variation in and Spatial Structure of Natural Populations of Dipterocarpus alatus (Dipterocarpaceae) Determined Using Single Sequence Repeat Markers. Genetics and Molecular Research 2014, 13, 5378–5386, doi:10.4238/2014.July.24.17.
66. Prajapati, S.; Sapkota, M.; Mishra, A.; Singh, S.; Sharma, T.; Panicker, L.; Lakshmi, S.M. An Overview of Forensic Analysis Methods of Pterocarpus santalinus L.f. and Their Suitability to Combat Biodiversity Crime, Adulteration and Environmental Exploitation: A Review. Discover Plants 2025, 2, doi:10.1007/s44372-025-00088-5.
Authors
Copyright (c) 2026 Nawwall Arrofaha, Henti Hendalastuti Rachmat, Fifi Gus Dwiyanti, Wahyu Catur Adinugroho, Iskandar Zulkarnaen Siregar, Irsyad Kamal, Dhika Syaputra, Andi Salamah

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.
- Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See The Effect of Open Access).